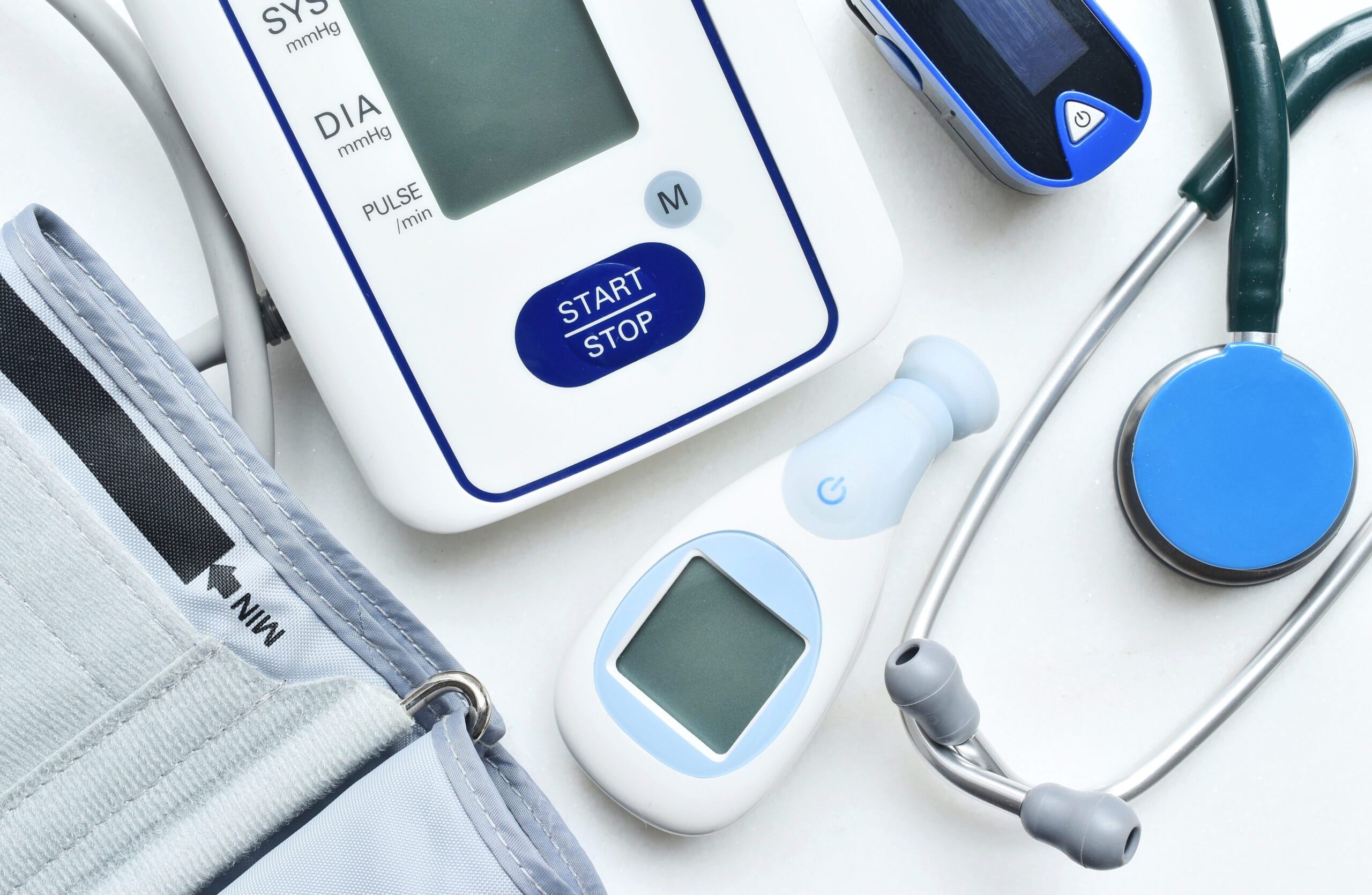
Clinical Devices & Equipment
Clinical and rehabilitation devices must deliver dependable performance under the most demanding conditions. Every component, actuator, housing, and cable plays a role in restoring movement and aiding recovery.

Medical and healthcare innovators partner with DTI because we understand the realities behind these expectations. We work closely with design, engineering, and quality teams from concept through to production, helping refine geometry, material selection, assembly strategy, and compliance considerations long before a device reaches testing or regulatory review. Whether supporting rehabilitation systems, mobility technologies, diagnostic equipment, prosthetic devices, or emerging wearable technologies, our approach reduces risk while strengthening long-term performance.
Our experience in ISO 13485-aligned manufacturing, FDA-relevant materials, and cleanroom-compatible tooling enables us to produce components that meet strict safety and traceability requirements. From first prototype to full-scale manufacture, we focus on delivering predictable behaviour, minimising waste, and ensuring every part contributes to a safe, dependable, and user-centred medical product.
By combining engineering precision with practical, real-world understanding, we help clients move from idea to certified production more quickly and with fewer obstacles. The result is devices that perform exactly as expected – supporting better care, empowering independence, and making meaningful differences to the lives they touch.

Clinical and rehabilitation devices must deliver dependable performance under the most demanding conditions. Every component, actuator, housing, and cable plays a role in restoring movement and aiding recovery.

Medical and laboratory consumables must deliver precision, hygiene, and compliance at scale, all while balancing cost control with increasing environmental responsibility.

Every component in a clinical device, from diagnostic sensors to prosthetic joints, must perform with absolute reliability while meeting strict global standards for safety, hygiene, and biocompatibility.

Whether you have a question, require more information, or want to discuss a potential project, our expert engineering and operations teams are on hand to help you.
We look forward to hearing from you and collaborating on innovative solutions for your business.
"*" indicates required fields